Lesson 7
Reasoning about Solving Equations (Part 1)
Problem 1
There is a proportional relationship between the volume of a sample of helium in liters and the mass of that sample in grams. If the mass of a sample is 5 grams, its volume is 28 liters. \((5, 28)\) is shown on the graph below.
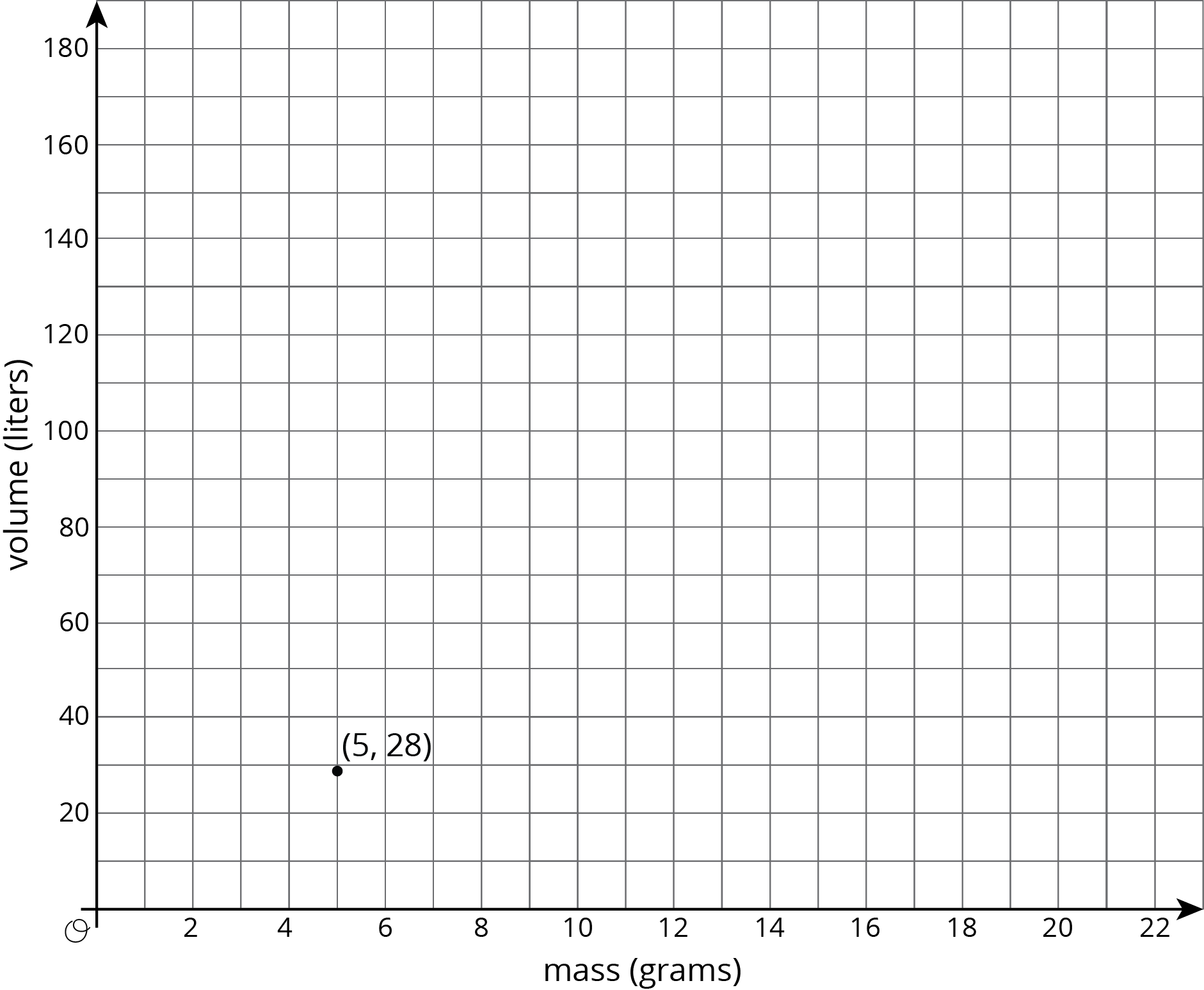
- What is the constant of proportionality in this relationship?
- In this situation, what is the meaning of the number you found in part a?
- Add at least three more points to the graph above, and label with their coordinates.
- Write an equation that shows the relationship between the mass of a sample of helium and its volume. Use \(m\) for mass and \(v\) for volume.
Solution
For access, consult one of our IM Certified Partners.
(From Unit 2, Lesson 11.)Problem 2
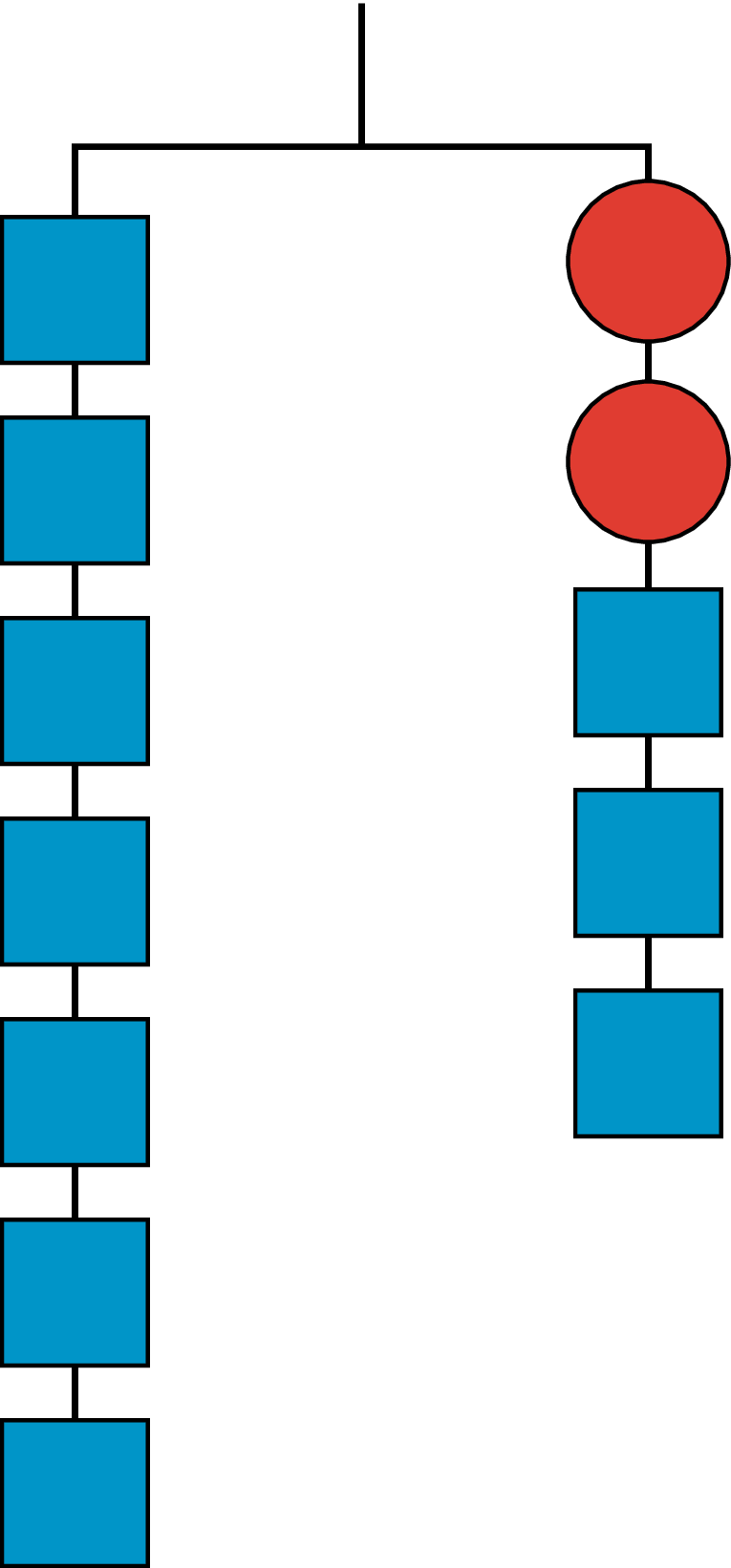
Explain how the parts of the balanced hanger compare to the parts of the equation.
\(7=2x+3\)
Solution
For access, consult one of our IM Certified Partners.
Problem 3
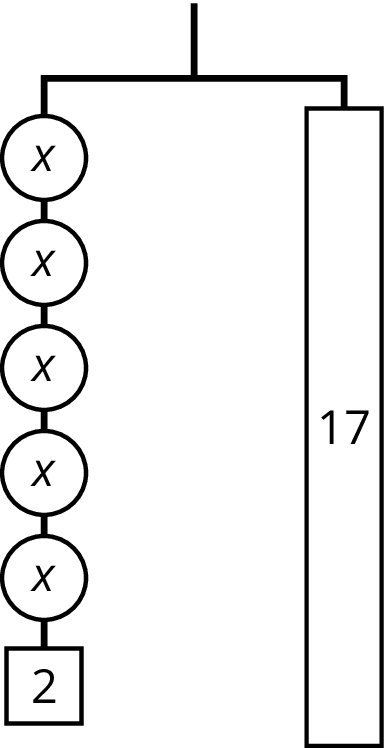
For the hanger below:
- Write an equation to represent the hanger.
- Draw more hangers to show each step you would take to find \(x\). Explain your reasoning.
- Write an equation to describe each hanger you drew. Describe how each equation matches its hanger.
Solution
For access, consult one of our IM Certified Partners.